HPLC Method for Nonpolar Molecules; How to Separate Naphthalene and Anthracene?
HPLC Method for Nonpolar Molecules: A Case Study: High-Performance Liquid Chromatography (HPLC) is an essential analytical technique in pharmaceutical development, particularly when dealing with the separation and quantification of compounds. However, developing an effective HPLC method requires a deep understanding of the underlying principles, especially when working with nonpolar compounds.
In this article, I will provide a skill-based guide to developing HPLC methods tailored to nonpolar compounds. By the end of this guide, you will gain the expertise to independently develop HPLC methods for nonpolar molecules. Additionally, you’ll be equipped to answer key questions related to the challenges and solutions in the process, ensuring a robust and reproducible method.
Non-Polar Molecules: When there is no separation of charge on the molecule then the molecule is called a Nonpolar molecule. Typical examples are Benzene, Naphthalene and Anthracene.
Principle of separation: Like interact like principle will govern the separation. Here Molecules are nonpolar and non-polar HPLC columns will be the suitable choice for the separation. Both Naphthalene and Anthracene are non polar compounds.
Since Naphthalene and Anthracene are non-polar compounds, a column containing non-polar stationary phase, like C18, C8, will be suitable for the separation.
These compounds can be separated either in gradient or isocratic mode, but gradient mode should be the preferred choice during method development. The trial should be started with a higher aqueous phase of 80%. Based on the elution of each Naphthalene Anthracene, optimisation should be done. Example:
| Time | A (water) | B (Methanol) |
| 0 | 80 | 20 |
| 15 | 20 | 80 |
| 20 | 20 | 80 |
| 20.1 | 80 | 20 |
| 25 | 80 | 20 |
Keep the flow rate between 0.5 to 1 ml/minute to get column pressure less than 2000 psi
Keep the sample concentration such that there should not be any column overloading, and the peak should be sharp. Sample concentration can be increased or decreased based on requirement e.g 0.2mg/ml, 0.5mg/ml, 0.7mg/ml
Keep the Injection Volume in such a so that there should not be any column overloading and the peak
should be sharp e.g 5μl, 10μl, 20μl.
Prepare the solution of each Naphthalene and Anthracene and scan in a UV spectrophotometer or PDA detector. Select a wavelength where each Naphthalene and Anthracene has almost an equal response
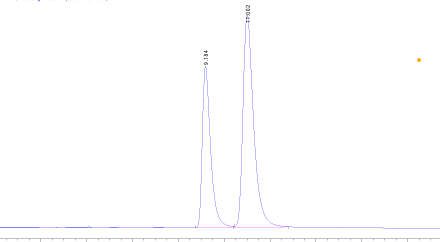
Inject the standard solution of each Naphthalene and Anthracene and generate the chromatogram. Inject the sample mixture and generate the chromatogram. Based on the elution pattern, optimise the mobile phase composition and chromatographic conditions to get better separation.
Based on the requirement, use area% % (area normalisation method) or the external standard method to give the result.
The molecular weight of Anthracene is higher than Naphthalene, and consequently, Anthracene is comparatively more nonpolar than Naphthalene. Therefore, Naphthalene will elute first and after that Anthracene will elute.
You have learned column section, mobile phase selection and chromatographic condition optimisation for nonpolar molecules in HPLC method development. I hope you can now develop the Analytical method for nonpolar compounds independently. Write your learnings/suggestions related to this post in the comment section.
Related:
To separate non-polar compounds, select non-polar columns like C18, C18 with high carbon load and a simple mobile phase like a mixture of water and organic solvent can be used.
Further Reading
Quick Links