What Is Product Validation and How Is It Performed? Learn With 7 FAQs
Product validation in pharmaceuticals explained—definition, types, importance, validation process, regulatory compliance, and FAQs.
Pharmaceutical Reference Standards: Primary Reference Standards, Secondary Working Standards & Applications (15 FAQs)
Learn about Pharmaceutical Reference Standards (PRS), including primary & secondary standards, applications, pharmacopeial sources, importance, and 15 FAQs for pharma professionals.
How to Avoid Analytical Errors in Pharmaceutical analysis: Easy 5-Minute Learning with FAQs & Case Study
Learn how to avoid analytical errors in pharmaceutical analysis with simple explanations, error types, prevention methods, FAQs, and a real QC case study.
Regulatory Compliance | 8 Common Mistakes, And How To Avoid Them Easily
Learn Regulatory Compliance in the Pharmaceutical Industry: 8 Common Mistakes Explained Simply with Case Studies and FAQs
Thermogravimetric Analysis (TGA) in Drug Development: Learn Easily with 7 FAQs and 3 Case Studies
Learn Thermogravimetric Analysis (TGA) in drug development with simple explanations, key principles, applications, FAQs, and real case studies.
Interview Questions on Humidity and Relative Humidity in Pharmaceuticals (QA, QC, Production & Validation)
Top interview questions on humidity and relative humidity in the pharmaceutical industry. Understand GMP requirements, RH limits, stability studies, and cleanroom control
Pharmaceutical Analysis in QC and ADL | Complete Guide to QMS, Calibration, Documentation, Troubleshooting & Analytical Techniques (HPLC, GC, MS, NMR, XRD, TLC, Titration, Spectroscopy)

Explore a complete guide to Pharmaceutical Analysis in QC and ADL, covering QMS, calibration, documentation, troubleshooting, and key analytical techniques like HPLC, GC, MS, NMR, XRD, TLC, titration, and spectroscopy.
Types of GC and HPLC Peaks: Learn With FAQs And Troubleshooting
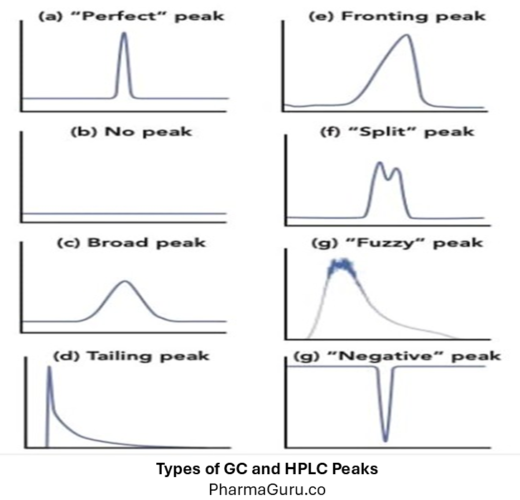
GC and HPLC peaks are graphical representations of compounds eluting from a chromatography system, where the x-axis denotes retention time, and the peak height or area corresponds to the compound’s concentration, providing both qualitative and quantitative information about the sample. Chromatography (both Gas Chromatography – GC and High-Performance Liquid Chromatography – HPLC) relies on the […]
Enantiomers, Diastereomers, Racemate and Meso-Form: Key Differences With Case Studies and FAQs
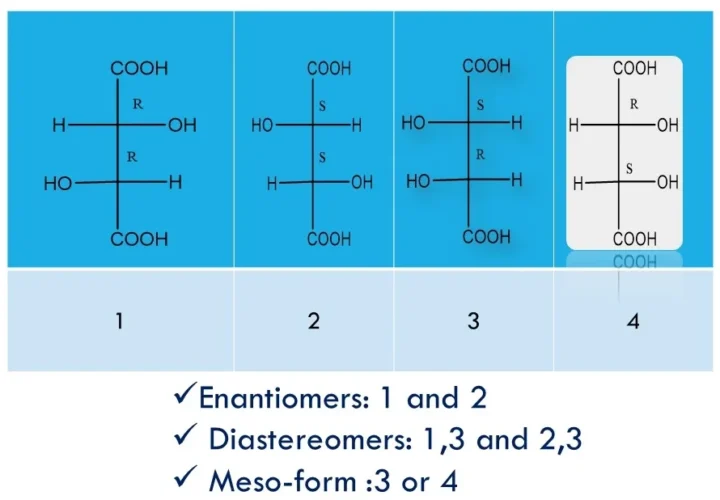
Enantiomers, diastereomers, racemates, and meso-compounds are types of stereoisomers—compounds with the same formula but different 3D arrangements—where enantiomers are non-superimposable mirror images with opposite chirality, diastereomers are non-mirror-image stereoisomers with different properties, racemates are 50:50 mixtures of enantiomers that are optically inactive, and meso-compounds have chiral centres yet are achiral due to internal symmetry. Enantiomers, […]
Swab Sampling and Rinse Sampling in Cleaning Validation: Learn In 3 Minutes
Swab sampling and Rinse sampling are key techniques in cleaning validation, each with distinct advantages. Guidelines recommend combining both methods for thorough residue assessment. Cleaning validation is a critical aspect of pharmaceutical manufacturing that ensures equipment and surfaces are free from residues of active pharmaceutical ingredients (APIs), cleaning agents, and microbial contaminants. Two primary sampling […]
