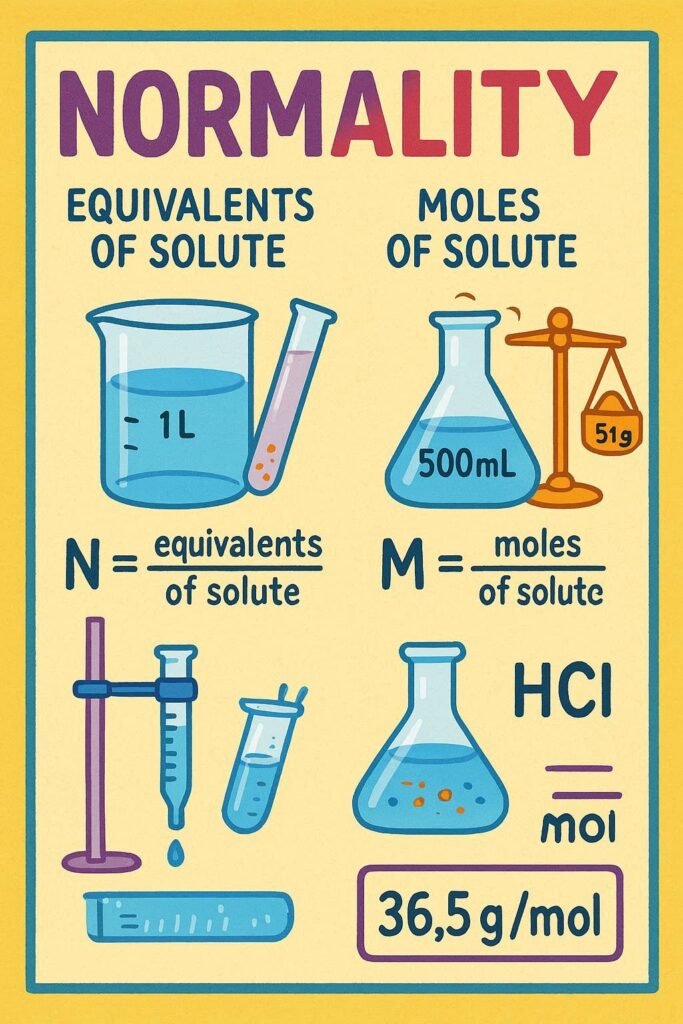
. Normality and Molarity measure solution concentration, but they’re not the same. Normality measures the concentration of reactive units (equivalents) per litre of solution, whereas Molarity measures the number of moles of solute per litre of solution What Is Difference Between Normality and Molarity? Feature Molarity (M) Normality (N) Definition Moles of solute per liter […]
.
Normality and Molarity measure solution concentration, but they’re not the same. Normality measures the concentration of reactive units (equivalents) per litre of solution, whereas Molarity measures the number of moles of solute per litre of solution
| Feature | Molarity (M) | Normality (N) |
|---|---|---|
| Definition | Moles of solute per liter of solution | Equivalents of solute per liter of solution |
| Depends On | Only the solute’s molecular formula | Type of reaction (acid-base, redox, etc.) |
| Reaction Specific? | No | Yes |
| Units | mol/L | eq/L |
| Example (H₂SO₄) | 1 M = 1 mol H₂SO₄/L | 1 M = 2 N (since H₂SO₄ donates 2 H⁺) |
Normality measures the number of equivalents per litre of solution. An equivalent is the amount of a substance that reacts with or supplies one mole of H⁺ ions (in acid-base reactions) or one mole of electrons (in redox reactions).
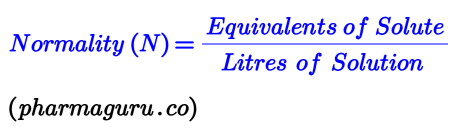
Note: Unlike molarity, normality depends on the type of chemical reaction. For instance:
Molarity is the number of moles of solute per litre of solution. It’s one of the most commonly used units of concentration in chemistry.
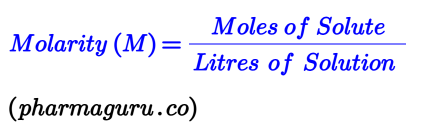
For example, if you dissolve 1 mole of NaCl in 1 litre of water, you get a 1 M NaCl solution.
Normality (N)=Molarity (M)×n-factor
While molarity tells you how many moles of a compound are in a liter, normality focuses on how many reactive units (equivalents) are involved. That’s why understanding the reaction type is crucial when deciding which unit to use.
Getting this distinction right ensures accurate lab results, efficient chemical calculations, and, ultimately, a better understanding of the reactions you’re working with.
Related Blogs:
Major Takeaway
Normality measures the concentration of reactive units (equivalents) per litre of solution, whereas Molarity measures the number of moles of solute per litre of solution.
Further Reading

Quick Links