Learn causes peak tailing and fronting in HPLC, and procedure to reduce them with FAQs
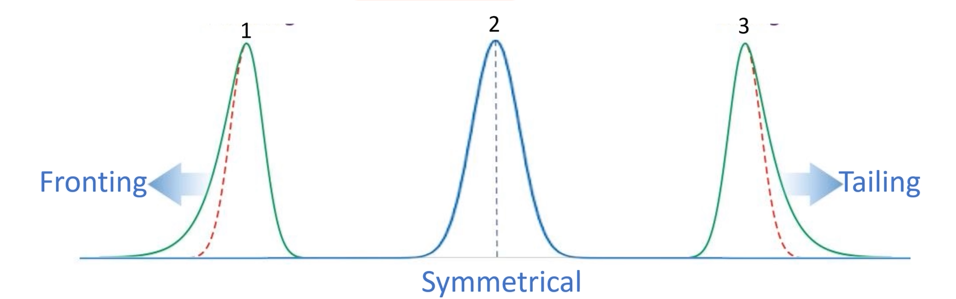
Tailing and Fronting are two common peak-shape issues that chromatographers face during HPLC and GC analysis. Peak tailing occurs when the peak skews toward the baseline after the apex- it stretches out and takes longer to return to baseline. In contrast, Peak fronting occurs when the peak skews to the left, rising too rapidly before the apex.
High-Performance Liquid Chromatography (HPLC) is a powerful tool used in analytical chemistry for separating, identifying, and quantifying compounds. However, achieving sharp, symmetrical peaks is crucial for accurate results. Two common peak shape issues analysts face are tailing and fronting. These distortions can affect resolution, quantification accuracy, and overall method robustness.
In this post, we’ll explore what causes peak tailing and fronting in HPLC, and how you can reduce them.

Peak tailing occurs when the peak skews toward the baseline after the apex- it stretches out and takes longer to return to baseline. This appears as a “tail” on the right side of the peak.
Peak fronting occurs when the peak skews to the left, rising too rapidly before the apex, with a gradual return to baseline.
| Aspect | Peak Tailing | Peak Fronting |
|---|---|---|
| Definition | Peak skews to the right (after apex), forming a ط “tail” | Peak skews to the left (before apex), rising too quickly |
| Peak Shape | Gradual return to baseline after apex | Sharp rise, gradual fall after apex |
| Main Cause | Strong or uneven interaction with stationary phase | Overloading or weak interaction with stationary phase |
| Column Overloading Type | Mass overload (too much analyte interacting) | Concentration overload (sample too concentrated) |
| Effect of Mobile Phase pH | Improper pH → partial ionization → strong interaction → tailing (common for bases) | Improper pH → inconsistent ionization → rapid/uneven elution |
| Stationary Phase Issues | Active sites (e.g., silanol groups, metal ions) cause adsorption | Column voids, poor packing, or compression issues |
| Column Condition | Contamination, aging → increased adsorption → tailing | Channeling or packing defects → uneven flow → fronting |
| Injection Issues | Incomplete injection or adsorption in injector | Over-injection or detector saturation |
| Flow Rate / Mobile Phase | Low flow or poor composition → longer interaction → tailing | High flow or strong solvent → rapid elution → fronting |
| GC-Specific Causes | Adsorption on active sites (Si–OH, metal oxides) | Thin film + overload or column defects |
| Common Compounds Affected | Polar/basic compounds (amines, alcohols) | Highly concentrated or fast-eluting compounds |
| Impact on Results | Poor quantification, reduced resolution | Distorted peak area, inaccurate quantification |
| Typical Acceptance | Tailing factor 0.9–1.5 acceptable; >2 problematic | Significant fronting usually unacceptable |
| Solutions | – Adjust pH – Use end-capped columns – Add modifiers (e.g., TEA) – Clean/replace column | – Reduce concentration/volume – Use higher capacity column – Replace poorly packed column |
| GC Column Role | Active sites & poor deactivation | Packing defects, film thickness issues |
1. Column Overloading
2. Poor Column Condition or Contamination
3. Inappropriate Mobile Phase pH
4. Active Sites on the Stationary Phase
5. Poor Injection Technique or Equipment Problems
6. Inappropriate Flow Rate or Mobile Phase Composition
7. Improper Column Packing
Routine system suitability tests (e.g., tailing factor, theoretical plates, resolution) can alert you early to method or system issues. A tailing factor (T) between 0.9–1.5 is generally acceptable; values beyond 2 may indicate a problem.

Most of the time, the GC column is responsible for tailing, fronting, and peaking.
Peak tailing often indicates unwanted interactions between the analyte and residual active sites such as exposed silanol groups (Si–OH) or metal oxides (e.g., Fe₂O₃, Al₂O₃) on the inner surface of the column. These sites can remain active when deactivation is incomplete or degrades over time or with elevated temperatures.
Tailing is particularly common with polar analytes (e.g., amines, acids, alcohols), and tends to be more pronounced when using nonpolar stationary phases, thin films, or analytes with low retention factors (k′), where a larger proportion of the analyte interacts with the column surface.
A broad peak indicates a loss of column efficiency, even when peak symmetry is maintained.
Primary column-related causes include:
These factors result in a wider range of analyte migration velocities through the column, leading to reduced theoretical plate count and compromised resolution. This behavior reflects the influence of the B (longitudinal diffusion) and C (mass transfer resistance) terms in the van Deemter equation.
Phase chemistry not only determines analyte retention but also significantly influences peak symmetry.
Film thickness also plays a critical role:
If your GC peaks exhibit unexpected asymmetry, excessive width, or unusual shapes—and the injector and detector are functioning properly—turn your attention to the column. Its surface chemistry, physical dimensions, and film characteristics play a direct role in governing analyte retention and peak shape.
| Observation | Likely Issue |
|---|---|
| Peaks getting worse over time | Column contamination or aging |
| Increased backpressure | Column blockage |
| Inconsistent retention time | Column or flow instability |
| Sharp leading edge | Fronting (overload) |
| Long trailing edge | Tailing (adsorption) |
Peak tailing and fronting aren’t just cosmetic issues—they directly affect method accuracy, reproducibility, and regulatory compliance. Understanding their root causes and applying targeted solutions can greatly improve the quality of your HPLC results.
Realted:
Major Takeaway | FAQs
Yes. If the pH is not optimised for the analyte’s pKa, it can result in partial ionisation and inconsistent interaction with the stationary phase, leading to tailing (often for basic compounds). Fronting can also result from poor ionisation control when analytes elute too rapidly or inconsistently
Not always. Mild tailing or fronting can sometimes be acceptable, especially if the peaks are well-resolved and quantification is not affected. However, significant asymmetry usually signals method development, sample, or hardware issues that should be addressed to ensure robustness and reproducibility.
Signs your column may be at fault include:
Further Reading

Quick Links