
Learn how to set up Identification test specifications for pharmaceuticals
An Identification Test in pharmaceutical analysis verifies that a drug substance or product contains the claimed active ingredient by comparing its properties to a known reference standard. Using techniques like spectroscopy, chromatography, and other physical or chemical methods, it plays a vital role in quality control by confirming authenticity, detecting impurities, and ensuring the safety and efficacy of the pharmaceutical.
Chromatography, such as Thin Layer Chromatography (TLC) or High-Performance Liquid Chromatography (HPLC), separates the components of a substance. The resulting chromatograms are compared to reference standards to confirm the identity of the substance based on its retention time or movement.
Spectroscopic methods like UV-Vis, IR, and NMR are used to obtain a unique spectral fingerprint of a substance. This helps identify the presence of specific functional groups or molecular structures, which can be compared to reference standards to confirm identity.
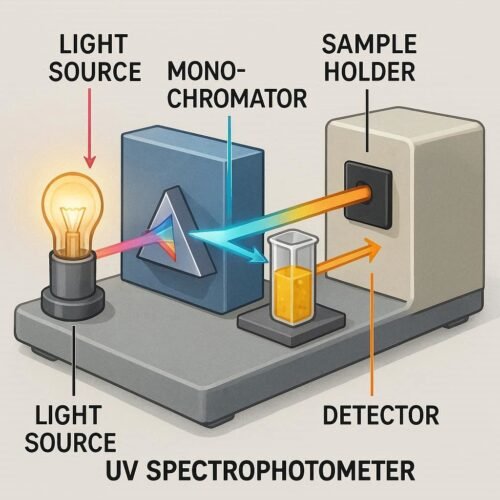
UV-Vis spectroscopy measures the absorption of ultraviolet and visible light by a substance. Each compound absorbs light at characteristic wavelengths, creating a unique absorption spectrum. By comparing this spectrum with a known standard, the substance can be identified.
Regulatory bodies like the United States Pharmacopoeia (USP), European Pharmacopoeia (EP), and International Council for Harmonisation (ICH) set standards for identification testing methods. These guidelines ensure that the tests are accurate, reliable, and reproducible for pharmaceutical substances.
Identification tests confirm the substance’s identity, while purity tests assess the level of contaminants or impurities in the substance. Both are critical for ensuring the quality and safety of pharmaceutical products.
Infrared (IR) spectroscopy identifies specific functional groups within a molecule by measuring the absorption of infrared light at various wavelengths. The resulting spectrum provides a “fingerprint” of the compound, which can be compared to a reference standard to confirm its identity.
Mass spectrometry (MS) identifies substances based on their mass-to-charge ratio. The resulting data can be used to determine the molecular structure and composition of a substance, allowing for precise identification of an API or pharmaceutical product.
Thin Layer Chromatography (TLC) separates compounds in a mixture by their affinity to a stationary phase. The pattern of spots formed on the TLC plate can be compared to a reference standard to confirm the identity of the substance.
An identification test in pharmaceutical analysis is a procedure used to confirm the identity of a pharmaceutical substance, such as an active pharmaceutical ingredient (API) or a drug product, to ensure it is as described and free from contamination or adulteration.
Pharmaceutical specifications define the quality attributes of a drug product, including its identity, purity, strength, and quality. The inclusion of identification tests in these specifications is essential to ensure that the drug substance or product is authentic. The key criteria for inclusion of identification tests in pharmaceutical specifications include:
There are several techniques used for identification tests in pharmaceutical analysis, and these can vary depending on the type of substance being tested and the required sensitivity and specificity. Below are some of the key techniques:
The infrared absorption spectrum of, pharmaceutical sample is concordant with the reference spectrum of the reference standard of the pharmaceutical
The analyte peaks in the chromatogram obtained with the test solution correspond to the analyte peaks in the
chromatogram obtained with the reference solution. In the following GC identification chromatogram each peak in the sample chromatogram is matched with its corresponding reference standard peak in the standard chromatogram

Retention time of the main pek in the sample chromatogram should match with the retention time of the desired isomer peak in the reference standard chromatogram
The Rf of the main spot in the sample solution should match with its correponding standard Rf in the standard solution
Identification tests offer several advantages, which are crucial for the safety and efficacy of pharmaceutical products:
Identification tests are a fundamental component of pharmaceutical analysis, ensuring that drugs are safe, effective, and meet regulatory standards. From chemical to spectroscopic methods, these tests are crucial in verifying the authenticity of substances used in drug development and production. By maintaining strict adherence to identification procedures, pharmaceutical manufacturers can protect consumers, improve product quality, and meet regulatory requirements, all while safeguarding public health.
The integration of reliable identification tests within pharmaceutical specifications contributes to high-quality manufacturing practices and ensures that the final products delivered to the market are safe and effective for patient use.
Related:
An identification test in pharmaceutical analysis is a procedure used to confirm the identity of a pharmaceutical substance, such as an active pharmaceutical ingredient (API) or a drug product, to ensure it is as described and free from contamination or adulteration.
These tests are crucial to ensure the quality, safety, and efficacy of pharmaceutical products. They help verify that the substance is correctly identified, authentic, and free from harmful impurities, preventing adverse effects in patients.
Yes, identification tests can help detect counterfeit drugs by verifying whether the substance matches the expected identity of the genuine product. Spectroscopic and chromatographic methods, in particular, are useful for identifying discrepancies in composition.
The identification test is important because it confirms the presence of the correct active pharmaceutical ingredient (API) in a drug, ensuring its authenticity, quality, and safety for patient use.
Identification of pharmaceutical impurities is the process of detecting and characterizing unwanted chemicals present in a drug substance or product, which may arise during manufacturing, storage, or degradation, to ensure product safety, quality, and regulatory compliance. Generally, LCMS, GCMS and NOR are used for identification of pharmaceutical impurities
The identification test for raw materials is performed to confirm the identity of each incoming material by comparing its physical or chemical properties to a reference standard, ensuring it is the correct substance before use in manufacturing.
Further Reading

Quick Links