PDA (Photo Diode Array) Detector/DAD is a widely used technique to check the HPLC peak purity in the pharmaceutical industry due to its simplicity and fast result. In this article, you will learn about the peak purity concept, different methods for calculating peak purity, advantages, limitations, and applications of peak purity. You will also learn […]
PDA (Photo Diode Array) Detector/DAD is a widely used technique to check the HPLC peak purity in the pharmaceutical industry due to its simplicity and fast result. In this article, you will learn about the peak purity concept, different methods for calculating peak purity, advantages, limitations, and applications of peak purity. You will also learn the concept of the PDA detector. After reading the article, your doubts will be cleared and you will be able to answer several questions related to peak purity and the PDA detector.
The process of assessing the spectral uniformity of a chromatographic peak is called peak purity.
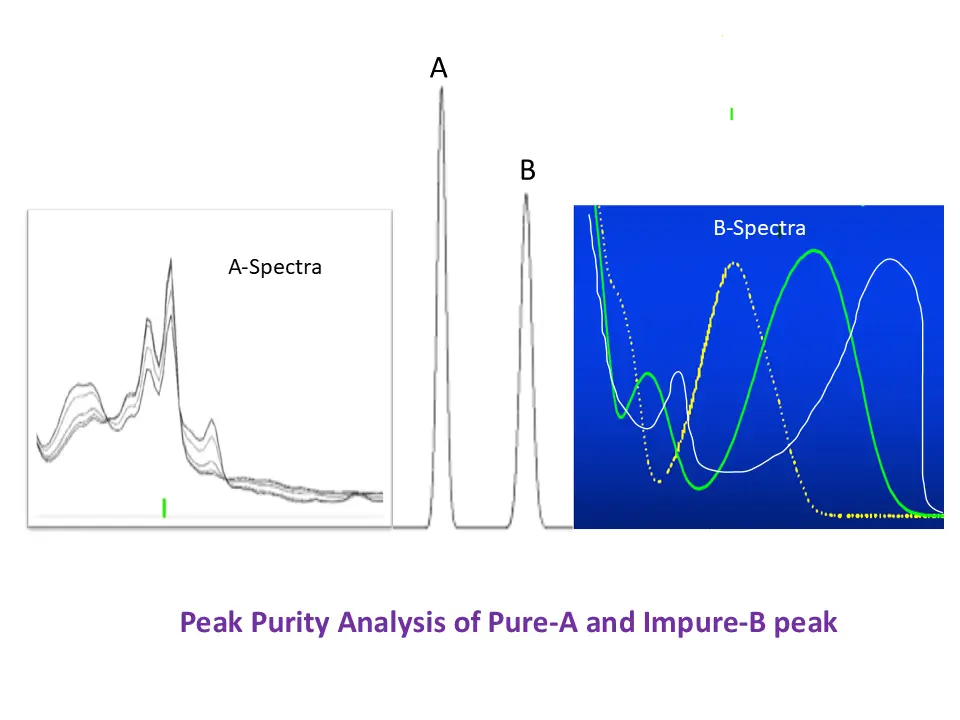
HPLC is used at almost all stages during pharmaceutical development to control the quality of APIs. It may be possible that other impurity peaks may co-elute with the peak of interest. Peak purity is performed to check the covariation/interference of other peaks with the peak of interest. In the following HPLC chromatogram (Figure1) impurity A and impurity B are coeluting with the main peak

Related: HPLC Method Adjustment Limits: What’s Allowed | FAQs and Case Studies
PDA detector or photodiode array detector acquires absorbance data over the entire uv-visible range (190 nm to 900 nm). Therefore an analytical scientist can extract chromatograms at multiple wavelengths within the selected wavelength range as per requirement during method development. Peak spectra can also be generated as per the required wavelength range. It is very helpful during HPLC method development. It is also called DAD or Diode array detector.
Most PDA instruments are available with wide UV-visible ranges such as 190 to 900nm. But some of the instruments may have narrow UV ranges like 190 nm to 650 nm or 200 nm to 650 nm
The following are the advantages of the PDA detector:
The following are the limitations of the PDA detector:
The process of assessing the spectral uniformity of a chromatographic peak is called the peak purity. This is the analysis of absorption spectra across peaks to determine whether they are all similar or have differences. It tells about the coelution of the peak in the main peak. This concept is based on the UV principle.
Peak purity by PDA detector is based on the comparison of spectra recorded during the elution of the peak (see Figure-1). Typically, the following three spectra are taken and compared to assess the peak purity by the PDA detector:
All the above three spectra satisfy the Peak purity suitability criteria.
Figure-2
One can select more than three points for spectra generation and can use all spectra for peak purity calculation.
In the figure-3:
Peak purity test may be affected:
Wavelength selection plays an important role in peak purity determination. If the wavelength has not been chosen scientifically then the peak purity result may be incorrect. The following factors must be considered while selecting the wavelength range:
| PDA | UV detector |
| In a UV detector, the operating wavelength (one or two as per instrument provision) is given and hencea chromatogram can be generated only at one or two wavelength | In a UV detector, the operating wavelength (one or two as per instrument provision) is given and hencea chromatogram can be generated only at one or two wavelengths |
| It can be used to determine both peak purity and chemical purity | It can only be used to determine chemical purity |
| Peak purity | Chemical purity |
| Peak purity tells about the homogeneity or purity of the chromatographic peak | Chemical purity tells about the chemical purity of the main molecule |
| It depends on those impurities which elute with the main peak. The impurities which are separated from the main peak can not affect the peak purity. | It depends on the impurities present in the molecules or each peak present in the chromatogram |
| It depends on noise, solvent composition, buffer concentration and co-elution of two of more than two peaks | It does not depend on noise, solvent composition and buffer concentration |
| It tells about the authenticity of the HPLC method | It tells about the quality/authenticity of the molecule |
| It is determined by HPLC, GC, Chemical titration, FTIR, NMR etc | It is determined by HPLC, GC, Chemical titration, FTIR, NMR etc |
The following techniques are widely used for peak purity:
Among the above methods, the spectral normalisation method and Spectral similarity method are widely used in industries for peak purity calculation
This technique involves normalising and comparing spectra taken across different points of the peak. This technique involves the following steps;
This technique very good for interactive data evaluation and for automated routine analysis
Figure-3
In this technique, all spectra from the concerned peak are compared with one or more selected spectra (reference spectra). Generally, the apex spectrum is taken as the reference spectrum. The degree of match or spectral similarity is plotted over time during elution.
The ideal profile for a pure peak is a flat line, and it is denoted as 1000. It is called a similarity curve.
At the start point and end point of each peak, spectra may vary due to the contribution of background noise take place. Due to this noise interference flat line is not always possible. That is the reason the threshold is kept. Generally, 980 is set as the threshold value for peak purity calculation.
The following criteria must be satisfied for peak purity:
The spectral comparison of the two spectra is called the match factor. The following are the relationships between the match factor and the spectra similarity:
The purity factor is the mean value of the match factor. It is the measure of the similarity in the shape of the spectra of a given peak.
The peak purity by the PDA detector is not a foolproof technique for peak purity checking of any peak. It has the following limitations and advantages:
The following are the limitations of the peak purity test:
The following are the advantages/applications of the peak purity test:
PDA is a basic technique to check HPLC peak homogeneity. It is also recommended that LC-MS be used while performing the peak purity.
HPLC peak purity assessment using a PDA/DAD detector is a fast and effective way to confirm chromatographic separation in pharmaceutical analysis. By understanding spectral comparison principles and the available purity calculation methods, analysts can quickly determine whether a peak represents a single compound or contains co-eluting impurities.
The PDA detector’s ability to capture full UV spectra across the peak makes it a practical and powerful tool during method development, validation, and routine QC. While not as selective as MS, its simplicity, speed, and accessibility make it ideal for everyday purity checks.
With the concepts explained in this article, you can now confidently evaluate peak purity using a PDA detector and apply this knowledge to strengthen the reliability of your HPLC methods.
Boost your pharma career with PharmaGuru’s expert-led online courses.: Online Pharma Course (Training)
PDA scan is 190 nm to 900 nm
The process of assessing the spectral uniformity of a chromatographic peak is called the peak purity.
Both are the same.
The Peak purity result will be pure since most of the chiral isomers have the same UV spectrum
Three methods; Spectral normalisation method, spectral similarity method and ratio gram generation method are widely used in calculating peak purity
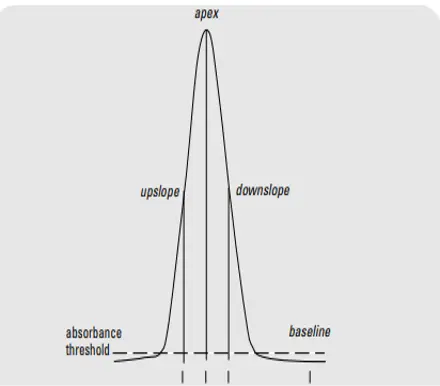
According to the purity concept a peak fails or passes the purity test depending on the matching of the spectrum of the peak on the upslope, peak maxima and downslope.
190 nm to 900 nm
PDA detector and mass detectors are used in HPLC for peak purity
The PDA detector acquires data simultaneously in UV and visible light. There the peak can be extracted at any wavelength of the wavelength range selected using the PDA detector.
The main difference between UV and PDA detectors is that UV detectors work at single wavelength, while the PDA detectors work at multiple wavelengths
The range of PDA is UV and visible light (190 nm to 900 nm).
For peak purity, spectra of at least three points upslope, peak maxima and downslope of any peak are taken and compared.A peak fails or passes the purity test depending on the match to the peak’s spectrum. Spectra of more than three points can also be collected and compared.
It tells about the co-elution of two or more peaks
In HPLC, a PDA detector is used to extract the chromatogram at multiple wavelengths in the single run
No
Quick Links